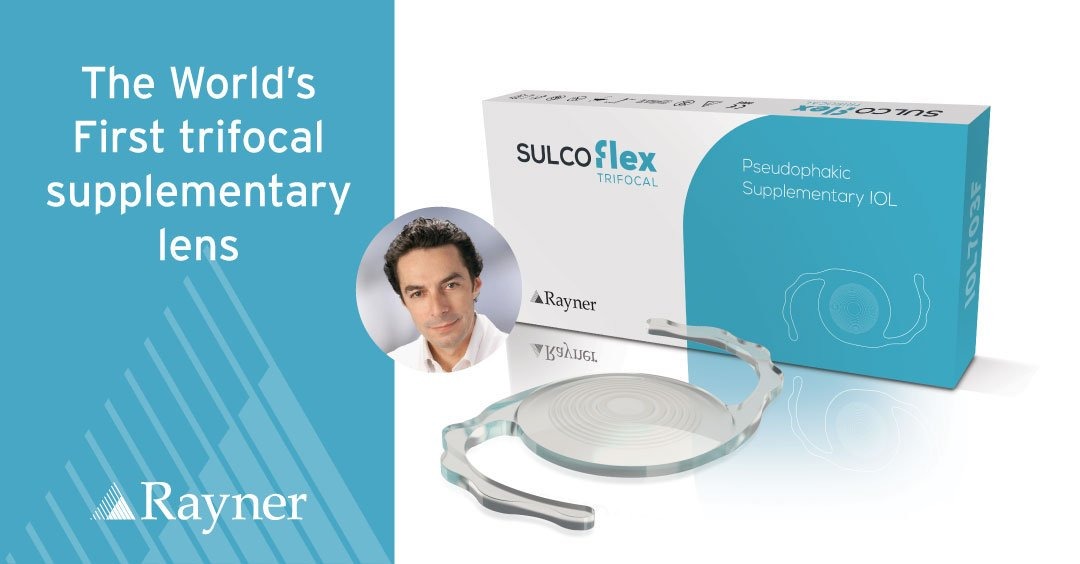
Rayner partners and acquires stake in Reper to drive R&D and build presence in Russia.
Posted on 1/09/2019
Rayner partners and acquires stake in Reper to drive R&D and build presence in Russia. Following a four-year R&D collaboration on a novel hydrophobic IOL material, Rayner has acquired a substantial stake in Reper, a leading material science and medical device company based in Russia. The new proprietary non-glistening material is the basis for RayOne